Valensa Yosephi; Peter Yustian Atmaja; Arif Nur Muhammad Ansori; Ahmad Affan Ali Murtadlo; Viol Dhea Kharisma; Putu Angga Wiradana; I Gede Widhiantara; Sukma Sahadewa; Maksim Rebezov; Nikolai Maksimiuk; Evgeniy Kolesnik; Marina Derkho; Natalia Koriagina; Aswin Rafif Khairullah; Fara Disa Durry; Rahadian Zainul
Abstract
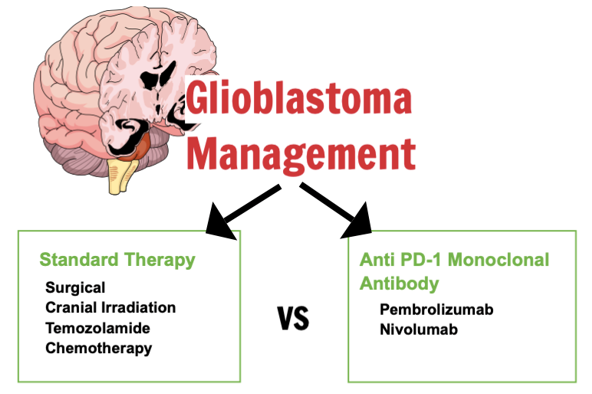
Monoclonal antibodies composed entirely of human immunoglobulin G4 that specifically target programmed death-1 (PD-1) are referred to as Nivolumab and Pembrolizumab. Monoclonal antibody ...
Read More
Monoclonal antibodies composed entirely of human immunoglobulin G4 that specifically target programmed death-1 (PD-1) are referred to as Nivolumab and Pembrolizumab. Monoclonal antibody of anti-PD-1 type for glioblastoma was found to be beneficial and harmless in preclinical research. Monoclonal antibody of anti-PD-1 (Pembrolizumab, Nivolumab) versus standard therapy for glioblastoma was systematically compared in terms of overall survival (OS), progression-free survival (PFS), and adverse events (AE). Pubmed, Scopus, Cochrane, and Clinicaltrials.gov databases were systematically searched through May 2023 for phase I or later RCTs reporting outcomes of interest. Inclusion RCTs criteria are adult population (>18 years old) with newly diagnosed or recurring cases of glioblastoma and have received immunotherapy with PD-1 inhibitors or standard treatment. For both OS and PFS, pooled effects estimates were computed using the approach of Cox proportional hazards regression. A computation was conducted on RCTs comprising 1680 patients from 4 included studies. Comparing anti-PD-1 monoclonal antibody to standard therapy, neither PFS nor OS improved at the median (HR = 1.08, 95% CI = 0.87-1.34, P = 0.51; P = 0.29; P = 0.29; and P = 0.29, 95% CI = 0.85-1.74, respectively). Furthermore, neither methylated nor un-methylated MGMT status benefits from anti-PD-1 monoclonal antibody (HR = 1.06, 95% CI = 0.89-1.27, P = 0.49; and HR = 1.29, 95% CI = 1.10-1.53, P = 0.002, respectively) in terms of overall survival. The anti-PD-1 antibody group experienced a greater incidence of AE (RR = 1.03, 95% CI = 0.94-1.14, P = 2.51). Anti-PD-1 monoclonal antibody did not demonstrate any clinical advantage over standard treatment for glioblastoma, according to this meta-analysis of four RCTs.